Measured Performance
Captured coverage maps for representative blood-borne pathogen panels.
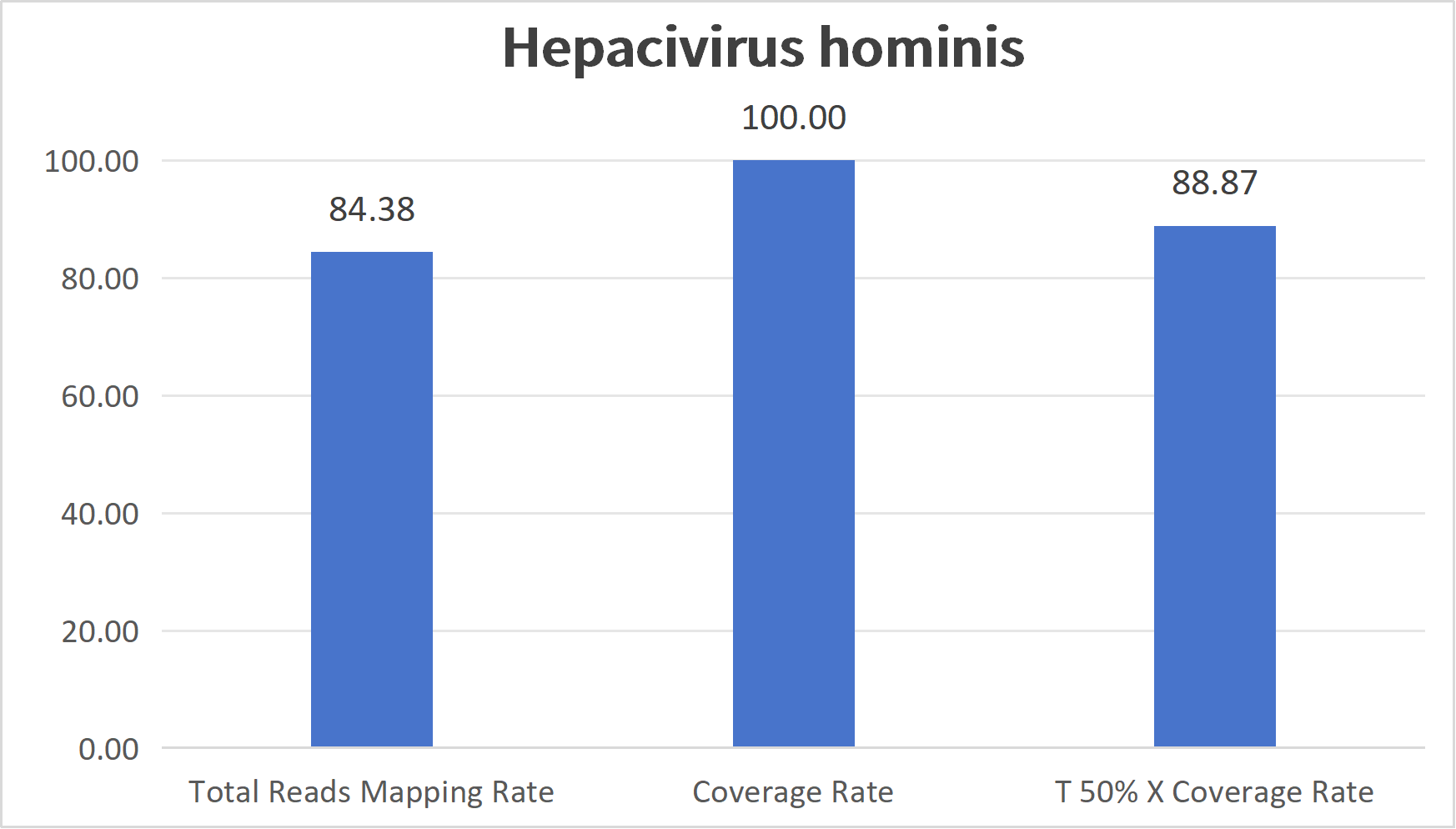
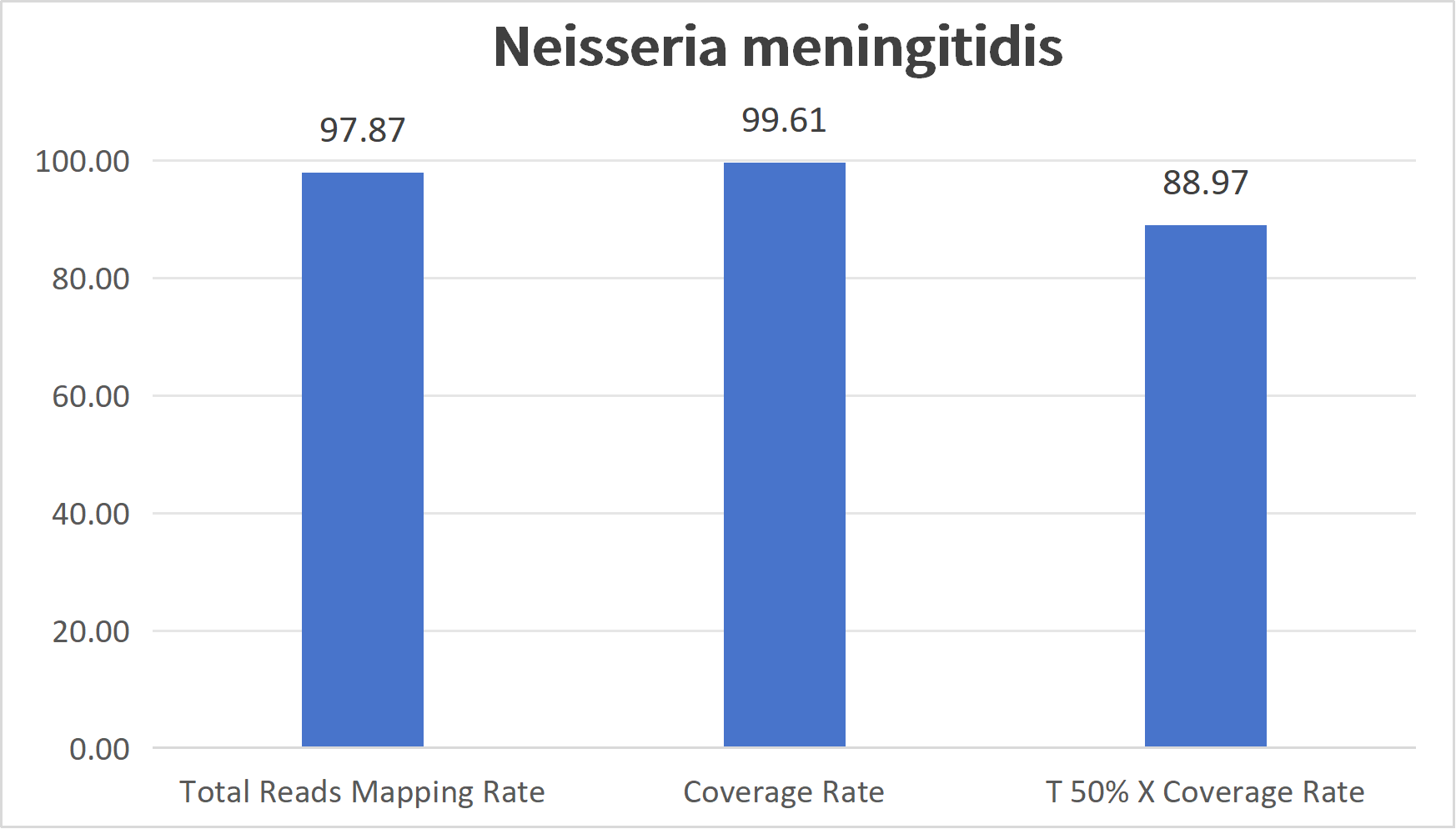
Blood related pathogens refer to microorganisms that can persist long-term in the human bloodstream and cause infectious diseases, covering viruses, bacteria, parasites, and other major types. Their transmission routes include blood transfusion and blood product infusion, shared contaminated syringes, mother-to-child transmission, and close contact with an infected person's body fluids. Infections often lead to chronic liver disease, immunodeficiency, sepsis, and other severe outcomes, and some of these diseases have extremely high mortality.
iGeneTech employs the proprietary TargetSeq® liquid-phase probe hybrid capture technology and bespoke microbial probe design to address critical challenges such as viral mutation monitoring, low-abundance viral sample detection, and host tracing, and now offers multiple whole-genome assay kits for blood related pathogens.
Selected panels covering bloodborne viruses and bacterial threats, with transparent reference metrics for each design.
| Product Name | English Name | Abbreviation | Taxonomy ID | Reference Genome Size | Reference Sequence Number | Number Of Probe |
|---|---|---|---|---|---|---|
| Hepacivirus Hominis Panel | Hepacivirus hominis | HCV | 3052230 | 9.4 kb | 1132 | 75136 |
| Lyssavirus Panel | Lyssavirus | LYSV | 11286 | 11.9 kb | 3469 | 43985 |
| Clostridium Botulinum Panel | Clostridium botulinum | C. botulinum | 1491 | 3.7 Mb | 74 | 645053 |
| Neisseria Meningitidis Panel | Neisseria meningitidis | N. meningitidis | 487 | 2.1 Mb | 144 | 122125 |
| Hepatitis A & E Virus Panel | Hepatitis A virus | HAV | 12092 | 7.5 kb | 142 | 120325 |
| Orthohepevirinae | / | 2946639 | 7.2 kb | 583 | ||
| Hepatitis Delta Virus Panel | Hepatitis delta virus | HDV | 12475 | 1.7 kb | 765 | 6871 |
Captured coverage maps for representative blood-borne pathogen panels.


Catalog SKUs for the highlighted bloodstream pathogen panels.
| Product Name | 16 rxn | 96 rxn | Order |
|---|---|---|---|
| Hepacivirus Hominis Panel | PH2003051 | PH2003052 | Order |
| Lyssavirus Panel | PH2006931 | PH2006932 | Order |
| Clostridium Botulinum Panel | PH2010931 | PH2010932 | Order |
| Neisseria Meningitidis Panel | PH2010921 | PH2010922 | Order |
| Hepatitis A & E Virus Panel | PH2011651 | PH2011652 | Order |
| Hepatitis Delta Virus Panel | PH2014261 | PH2014262 | Order |
The Blood Related Pathogens Panel enables simultaneous detection and identification of over 70 bloodstream pathogens from a single sample, covering bacteria, viruses, fungi, and parasites that cause life-threatening bloodstream infections.
Targets pathogens commonly causing bacteremia, sepsis, and other bloodstream infections
Detects bacteria, viruses, fungi, and parasites from blood samples
Complete genome sequencing enables strain typing and resistance gene detection
Fast workflow delivers results within 24 hours for timely sepsis management
Advanced technology for bloodstream pathogen detection
Detects over 70 bloodstream pathogens including rare and emerging threats
Full-genome analysis identifies antibiotic resistance mechanisms
Detects low-level pathogens and unculturable organisms
Identifies multiple concurrent bloodstream infections
Compatible with automated platforms for consistent high-throughput testing
Results within 24 hours for rapid clinical decision making
Key literature supporting the development and evaluation of the blood pathogen panels.
Contact our team to learn more about the Blood Related Pathogens Panel and how it can benefit your laboratory or clinical facility.